The Biopharma SHAKTI Revolution
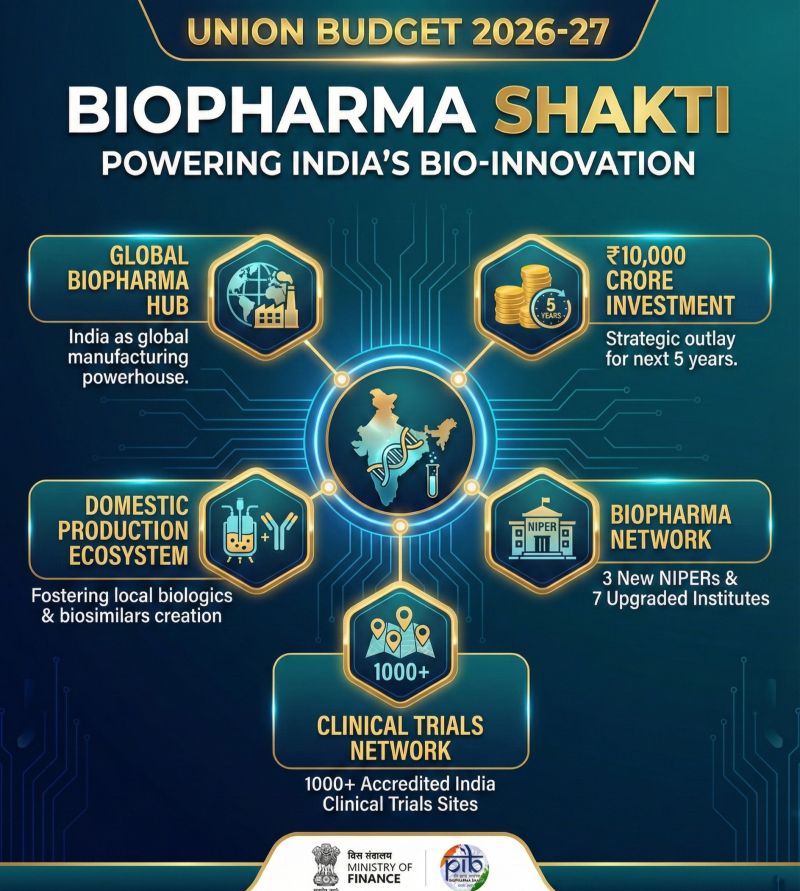
The Biopharma SHAKTI mission and stock beneficiaries (Strategy for Healthcare Advancement through Knowledge, Technology, and Innovation) mission is the “Big Bang” moment for India’s life sciences. Announced in the Union Budget 2026 with a massive ₹10,000 crore outlay, this 5-year initiative is designed to pivot India from a generic “pill-maker” to a global “biologics innovator.”
Unlike traditional chemical drugs, biologics are grown from living cells and are essential for treating Non-Communicable Diseases (NCDs) like cancer and autoimmune disorders. The mission establishes 3 new NIPERs, upgrades 7 existing institutes, and—most importantly—creates a powerhouse network of 1000 accredited clinical trial sites. By strengthening the CDSCO with a dedicated scientific review cadre, India aims to match global FDA approval timelines, capturing 5% of the $110 billion global biologics market.
What is the Biopharma SHAKTI mission in India?
The Biopharma SHAKTI (Strategy for Healthcare Advancement through Knowledge, Technology, and Innovation) mission is a ₹10,000 crore Indian government initiative announced in the Union Budget 2026. It aims to transform India into a global manufacturing hub for biologics and biosimilars.
The Breakdown: Biopharma SHAKTI Mission Details and Stock Beneficiaries
| Feature | Details of the SHAKTI Mission |
| Financial Outlay | ₹10,000 Crore over 5 years (2026–2031). |
| The “Triple Crown” | Focus on R&D, Industrial Entrepreneurship, and Biomanufacturing. |
| Stairway to Scale | Establishment of 3 new NIPERs and upgrading 7 existing ones into “Bio-innovation Hubs.” |
| Clinical Strength | Creation of 1,000 accredited clinical trial sites India list to stop “data flight” to other countries. |
| Regulatory Fast-Track | CDSCO will get a “Scientific Review Cadre” to cut drug approval times by 40%. |
| Disease Focus | Shift from infectious diseases to NCDs (Cancer, Diabetes, Autoimmune). |
The Core Objective
The primary goal is to capture 5% of the global biopharmaceutical market share by 2030.
- The Problem: India currently dominates “Small Molecules” (pills/generics), but the world is moving toward “Big Molecules” (Biologics).
- The Solution: SHAKTI provides the funding to build the labs and factories needed for Biologics and Biosimilars—medicines derived from living organisms rather than chemicals.
What is the difference between biologics and traditional drugs?
Traditional drugs are “small molecules” created through chemical synthesis (like aspirin). Biologics are complex “large molecules” manufactured from living organisms, cells, or tissues. They are highly effective for treating chronic conditions but require advanced manufacturing infrastructure.
Financial & Structural Outlay
- Total Budget: ₹10,000 Crore over 5 years.
- Focus Area: Non-communicable diseases (NCDs) like Cancer, Diabetes, and Autoimmune disorders.
- New Infrastructure: * 3 New NIPERs: Establishing new National Institutes of Pharmaceutical Education and Research.
- 7 Upgrades: Upgrading existing NIPERs to world-class “Bio-Research Centres.”
- 1,000 accredited Clinical Trial Sites: Creating a massive, accredited network to speed up drug testing within India.
Biopharma SHAKTI mission and stock beneficiaries: The ₹10,000 Crore Blueprint
The Indian pharmaceutical landscape is undergoing a “tectonic shift.” With the announcement of the Biopharma SHAKTI mission and stock beneficiaries, it is clear that the government is moving beyond generic manufacturing. This ₹10000 crore Biopharma SHAKTI outlay initiative is the backbone of India’s bid to capture 5% of the global biologics market by 2030.
₹10,000 crore Biopharma SHAKTI outlay impact analysis
The ₹10,000 crore Biopharma SHAKTI outlay impact analysis reveals a multi-layered strategy. Unlike previous schemes, SHAKTI focuses on “Big Molecules.” While traditional chemical drugs (Small Molecules) have been India’s forte, the future lies in Biologics—complex medicines derived from living organisms.
This outlay isn’t just a subsidy; it’s an infrastructure overhaul. It includes the establishment of three new NIPERs and the transformation of seven existing ones into “Bio-Innovation Hubs.” For investors, this signals a massive shift in capital towards companies with high R&D capabilities. Much like the India IT Rules 2026 Business Impact reshaped digital compliance, SHAKTI is redefining pharmaceutical standards.
- External Resource: Read the official PIB release on India’s Bio-Economy 2026.
Top Indian biosimilar stocks for 2026 budget
Identifying the top Indian biosimilar stocks for 2026 budget is crucial for portfolio rotation. The SHAKTI mission provides weighted R&D tax deductions of up to 200%, making “Deep-Tech” pharma highly attractive.
- Biocon Ltd: The primary beneficiary due to its massive biologics pipeline.
- Sun Pharma: Shifting focus from generics to specialty oncology.
- Syngene International: The “Pick and Shovel” play that will manage clinical data.
Investing in these stocks requires the same discipline as learning how to start stock market trading in India 2026. The long-term gains in Biopharma are expected to outperform traditional sectors over the next five years.
What diseases does the Biopharma SHAKTI scheme target?
The initiative specifically targets the rising burden of Non-Communicable Diseases (NCDs) in India. Funding and research focus heavily on advanced treatments for cancer, diabetes, rare genetic conditions, and autoimmune disorders.
CDSCO scientific review cadre impact on pharma approvals
One of the most overlooked “Details” in the SHAKTI mission is the CDSCO scientific review cadre impact on pharma approvals. Historically, Indian drug approvals took 18–24 months. The mission introduces a dedicated team of specialized scientists within the Central Drugs Standard Control Organization (CDSCO) to cut this timeline down to 6–9 months.
This regulatory speed is a “Force Multiplier.” It allows companies to bring “First-to-Market” biosimilars to the global stage faster. This efficiency is as vital to a company’s bottom line as understanding the Section 87A rebate on special rate income is to an individual’s tax planning.
- External Resource: Check the latest CDSCO Regulatory Guidelines.
1000 accredited clinical trial sites
The mission’s promise of a 1,000 accredited clinical trial sites India list is designed to stop “data flight.” By providing localized, world-class testing grounds, the government ensures that clinical data for Indian populations stays within the country.
These sites will be equipped with high-end tech, likely monitored by the latest WiFi security cameras for home and labs to ensure 24/7 compliance and security. This network will significantly lower the “Cost of Innovation” for Indian startups.
Why is India shifting focus from generic drugs to biosimilars?
While India dominates the global volume of generic pills, the global market value is rapidly shifting toward biologics (projected to outpace chemical drugs by 2027). Biosimilars offer much higher profit margins and better treatments for complex diseases.
The “Big 5” Stock Beneficiaries
These companies are specifically positioned to capture the ₹10,000 crore outlay due to their existing biologics infrastructure.
- Biocon Ltd (BIOCN): The undisputed leader. With their global biosimilar portfolio and existing GLP-1 (weight loss drug) pipeline, they are the primary beneficiary of the new 1,000 accredited clinical trial sites India list network.
- Syngene International: As a top-tier CRDMO (Contract Research and Development Organization), they will be the “pick and shovel” provider for global companies setting up R&D under the SHAKTI mission.
- Sun Pharmaceutical Industries: They are aggressively moving away from generics into “Specialty Medicines.” The SHAKTI incentives for NCDs (Cancer/Diabetes) align perfectly with their 2026-2030 roadmap.
- Concord Biotech: Specialists in fermentation-based biopharma APIs. As India reduces import dependence on fermentation products, Concord is the structural winner.
- Piramal Pharma: Their focus on complex hospital generics and contract manufacturing makes them a prime candidate for the new “Integrated Biomanufacturing Parks.”
Biopharma SHAKTI vs National Biopharma Mission i3
When comparing the Biopharma SHAKTI vs National Biopharma Mission i3, the difference is scale and speed. While the 2017 “i3” mission focused on foundational research, SHAKTI is about Industrialization.
The i3 mission laid the tracks; SHAKTI is the high-speed train. It moves the focus from “discovery” to “commercialization at scale.” It is the difference between a solo content creator setup and a full-scale media production house.
What are the specific Biopharma SHAKTI mission details and stock beneficiaries?
The Biopharma SHAKTI mission details and stock beneficiaries revolve around a ₹10,000 crore strategic push into biologics. Key details include the creation of 1,000+ trial sites and the upgradation of NIPER institutes. Primary stock beneficiaries are Biocon, Syngene, and Sun Pharma, which hold the largest R&D pipelines for complex biosimilars in India.
What does the ₹10,000 crore Biopharma SHAKTI outlay impact analysis suggest for investors?
A ₹10,000 crore Biopharma SHAKTI outlay impact analysis suggests a major shift from volume to value. For investors, this means lower gestation periods for drug launches and reduced capital expenditure due to government-funded “Bio-innovation Hubs.” This outlay is expected to boost the sector’s CAGR from 12% to over 20% by 2027.
Which are the best biosimilar stocks India 2026 Budget has highlighted?
The best biosimilar stocks India 2026 Budget highlights are those with established fermentation and monoclonal antibody (mAb) platforms. Biocon Biologics remains the top pick, followed by Concord Biotech (API specialist) and Dr. Reddy’s. These companies are directly aligned with the SHAKTI goal of capturing 5% of the global biologics market.
How will the CDSCO scientific review cadre impact on pharma approvals work?
The CDSCO scientific review cadre impact on pharma approvals will be felt through significantly faster market authorization. By hiring specialized bioprocess engineers and scientists, the CDSCO aims to reduce the approval cycle for complex biologicals from 18 months to under 9 months, allowing Indian firms to compete with US and EU timelines.
Why is the 1,000 accredited clinical trial sites India list a game-changer?
The 1,000 accredited clinical trial sites India list is a game-changer because it provides a standardized, ready-to-use infrastructure for deep-tech startups. Instead of searching for hospitals, firms can access this network to conduct Phase I-III trials locally, drastically lowering the cost of drug development for Indian NCDs like cancer and diabetes.
What is the total budget outlay for Biopharma SHAKTI?
The Indian government has allocated a total outlay of ₹10,000 crore for the Biopharma SHAKTI mission. This funding will be distributed over five years (2026–2031) to build infrastructure, upgrade research facilities, and streamline regulatory approvals.
Which stocks will benefit most from the Biopharma SHAKTI scheme?
Top stock beneficiaries include Biocon, Syngene International, Sun Pharma, and Dr. Reddy’s Laboratories. Companies with strong existing pipelines in complex biologics, oncology, and contract research (CDMO) are best positioned to leverage the government’s new clinical trial networks and R&D incentives.
How does the Biopharma SHAKTI mission affect the CDSCO?
The mission significantly strengthens the Central Drugs Standard Control Organisation (CDSCO) by introducing a dedicated “Scientific Review Cadre.” This specialized team aims to align India’s regulatory approval timelines with global standards (like the US FDA), reducing drug approval wait times.
How many clinical trial sites will be built under the 2026 Budget?
The Biopharma SHAKTI initiative funds the creation of a nationwide network of over 1,000 accredited clinical drug trial sites. This ensures that clinical research data on Indian populations remains within the country and accelerates domestic drug development.
How many NIPER institutes are planned under the 2026 budget?
The government will establish 3 new National Institutes of Pharmaceutical Education and Research (NIPERs) and upgrade 7 existing institutes. These will serve as advanced “Bio-Innovation Hubs” to align academic research with global industry requirements.
Is Biopharma SHAKTI different from the National Biopharma Mission i3?
Yes. While the 2017 National Biopharma Mission (i3) had a ₹1,500 crore outlay focused on early-stage R&D and vaccines, the 2026 SHAKTI mission is a massive ₹10,000 crore industrialization push focused on scaling commercial manufacturing and global exports.
How will Biopharma SHAKTI impact the Indian economy?
By reducing India’s reliance on imported active pharmaceutical ingredients (APIs) and high-value biologics, the mission aims to capture 5% of the global biopharmaceutical market by 2030, contributing significantly to India’s target of a $300 billion bioeconomy.
What tax and PLI incentives are linked to Biopharma SHAKTI?
The mission complements existing Production Linked Incentive (PLI) schemes and offers weighted R&D tax deductions. Furthermore, the 2026 budget provides specific customs duty exemptions for importing specialized equipment needed for biomanufacturing.
How does the Biopharma SHAKTI mission help Indian startups?
Startups will benefit from the “plug-and-play” infrastructure of the new bio-manufacturing clusters and access to the 1,000+ clinical trial network, significantly lowering the capital needed to test and launch new biologic therapies.
Will the Biopharma SHAKTI mission create healthcare jobs?
Yes. The massive infrastructure expansion across NIPERs, CDSCO, and new clinical trial sites will create thousands of high-paying jobs for specialized scientists, clinical researchers, regulatory affairs experts, and advanced laboratory technicians.
Can foreign investors participate in the Biopharma SHAKTI initiative?
Absolutely. The government is actively courting foreign direct investment (FDI) and technology partnerships—such as recent sovereign fund pitches in Europe—to co-develop specialized equipment, full-stack design, and resilient supply chains in India.






